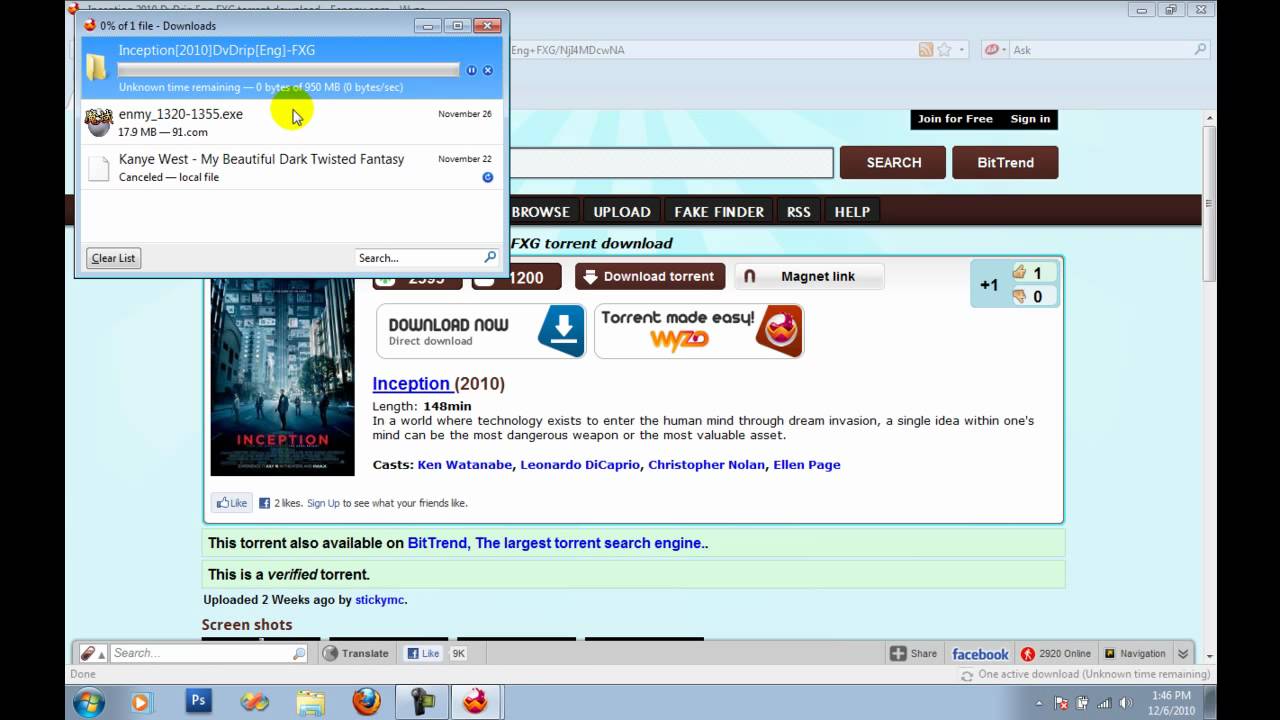
International Standards are drafted in accordance with the rules given in the ISO/IEC Directives, Part 2. The main task of the joint technical committee is to. Explore the world of DIN! In our video, we show our fields of activity and address important future topics. One thing for sure: it is with standards that we can create a future worth living together. Torrent Din Standards Free. 2/23/2017 0 Comments. Din standards torrent free download - din Standard Torrent, mitcalc 2010 for solidworks torrent. Download papers, chapters, and e-books NOW STANDARDS CATALOG www.astm.org. New Full-Text Search and Digital Object Identifier (DOI) Enhance Standards Searching. IHS Markit is your source for US and international engineering and technical standards, specifications, codes, and training materials in hardcopy of PDF download.
Free Download: IEC 60601-1 Compliance Documents
The following information and document downloads are tools to evaluate medical electrical equipment to the applicable standards.
They are provided for free to aid you in your medical equipment design and development, where this information is most useful.
Din Download Free
Dexis software manual. Please contact us if you have any questions or are interested in having us handle your product’s safety certification.
Medical Standards List with Publish Dates
- (Note, this document was revised on the specified date, so may not be current)
- Harmonized Standards with Publish Dates (Collaterals, Particulars)
- Anticipated publish dates for new standards, editions, amendments
- To find the current status and issue dates of these standards, see the Links Page, Standards
- To find and download full standards (which are copyrighted), please see the Resources page, Purchasing Standards
MECA 60601-1 Ed. 3.1 Evaluation Package (BETA)
- The Evaluation Package is a summary of the IEC 60601-1:2012 standard, other applicable requirements, guidance information, and interpretations, to help evaluate medical electrical equipment to the requirements of the Standard.
- It is being provided FREE of charge, to help people understand and meet the requirements for medical devices.
- The Evaluation Package is not intended to replace the standard, so a purchased copy should also be used.
- NOTE – This document is still under development, and only pages 1-37 are being provided at this time.
- It will be updated here as it is completed.
Risk Management Guidance Form for ISO 14971
ISO 14971 MECA F 027b – ISO 14971 Client Completion Form (0.2Revision)
- Covers Clause 4.2.2 of IEC 60601-1, Ed. 3.1, which is ISO 14971, Clauses 3.1 – 8
- Clients who have an evaluation project with MECA receive this in an editable Word format to complete
Din Standards Free
Risk Management Guidance Form for IEC 60601-1
IEC 60601-1 MECA F 028c – IEC 60601-1 2012 Risk Management Client Completion Form (0.2Revision)
- Covers Risk Management referenced Clauses in IEC 60601-1, Ed. 3.1
- Clients who have an evaluation project with MECA receive this in an editable Word format to complete
IEC 60601-1, Edition 3.1 Label-Manual Checklist
MECA IEC 60601-1 Ed3.1 Label-Manual Checklist Rev4.pdf (2015-01-28) Crack for autocad 2010.
- Checklist for the requirements of the Labelling and Accompanying documents
Medical Alarm Standards Cross-Reference
Burnout paradise pc serial key. meca-alarm-standards-cross-reference20150401.pdf (2015-04-01) https://formbrown345.weebly.com/blog/high-yield-obstetrics-and-gynecology-ebook.
- A comparative resource for those who work with alarm systems and the corresponding standards in the field of medical electrical equipment.
- Note, IEC authorization has been received to publish wording from these standards.
Medical Standards Project Scope Tool
MECA 60601-80601 Medical Standards Project Scope Tool (Rev 2015-08-17).pdf (2015-08-17)
- A list of IEC 60601 and the IEC/ISO 80601 Collateral and Particular standards, with their referenced and required additional standards and requirements.
- This is a great tool to be sure you’ve included everything when determining the applicable standards and requirements.
International Din Standard
IEC 60601-1 Critical Components Guidance Documents
- Critical Component Guide for IEC 60601-1
- This guidance document will aid you in identifying your device’s critical components and what specifications they may need to meet for documentation in your Compliance Report.
MECA 60601-1 Ed. 2 with National Differences Evaluation Package
- The Evaluation package is a summary of the IEC 60601-1:1988 + A1:1991 + A2:1995 standard, project process, national differences, and guidance information to help evaluate medical electrical equipment to the requirements of the Standard.
- It is being provided FREE of charge, to help people understand and meet the requirements for medical devices.
- The Evaluation Package is not intended to replace the standard, so a purchased copy should also be used.
